For this STEM project we were instructed to build a board game that included chemical reactions. We had to include one physical change (such as a liquid to a gas, solid to a liquid, solid to a gas, etc.), a single displacement reaction, a double displacement reaction, a chemical change, and the lighting of an LED light. This project was really fun and I really enjoyed working on it.
For our physical change we used dry ice and boiling hot water. When the dry ice touched the boiling hot water it turned from a solid form to a gaseous form.
For our single displacement reaction we used copper chloride and aluminum. The aluminum resists the LED from lighting up so when you put the copper chloride in it breaks down the aluminum and with it broken up the LED lights up. This served as both our lighting of our LED and our single displacement reaction.
For our double displacement reaction we used vinegar or acetic acid and mixed it with sodium bicarbonate more commonly known as baking soda. We put a balloon over the flask and let the gas fill up the balloon emulating a hot air balloon.
For our chemical reaction we made elephant toothpaste. In order to make this we used potassium iodide, dish soap, food coloring and 30% hydrogen peroxide. This experiment was my favorite.
For our physical change we used dry ice and boiling hot water. When the dry ice touched the boiling hot water it turned from a solid form to a gaseous form.
For our single displacement reaction we used copper chloride and aluminum. The aluminum resists the LED from lighting up so when you put the copper chloride in it breaks down the aluminum and with it broken up the LED lights up. This served as both our lighting of our LED and our single displacement reaction.
For our double displacement reaction we used vinegar or acetic acid and mixed it with sodium bicarbonate more commonly known as baking soda. We put a balloon over the flask and let the gas fill up the balloon emulating a hot air balloon.
For our chemical reaction we made elephant toothpaste. In order to make this we used potassium iodide, dish soap, food coloring and 30% hydrogen peroxide. This experiment was my favorite.
This is a picture of our finished board game!
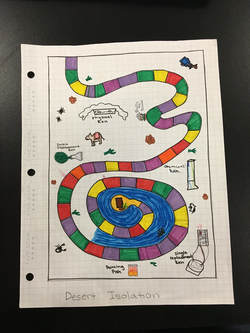
This is a picture of the blueprint of our project!
Safety Precautions
***DO NOT CONSUME ANY OF THE MATERIALS USED IN THESE EXPERIMENTS***
Wear protective gloves, goggles, and apron while all experiments are performed.
If chemicals contact your skin, rinse it in a nearby sink.
If chemicals get in your eyes, use a nearby fountain to flush it out.
If eyes or skin become irritated during experiment, rinse them with water.
If on fire or covered in large quantities of chemicals, use the safety shower.
Single Replacement Reaction: Wear protective gloves, apron, and goggles
This is the chemistry of all of our experiments within our board game:
Double Displacement Reaction: sodium bicarbonate (baking soda) and dilute acetic acid (vinegar)
NaHCO3(s) + CH3COOH(l) → CO2(g) + H2O(l) + Na+(aq) + CH3COO-(aq)
A double displacement reaction is where two aqueous ionic compounds exchange ions and form two new ionic compounds. It goes from two aqueous and aqueous to aqueous and either a solid, liquid, or gas. When this happens, it forms a precipitate. Acetic acid and sodium bicarbonate react in a double displacement reaction to form sodium acetate and carbonic acid. The carbonic acid then has a decomposition reaction and produces carbon dioxide gas which causes the solutions to bubble. Water and oxygen are also produced from this reaction. The release of the carbon dioxide gas flows out of the flask and into the balloon, inflating it.
Single Replacement Reaction: Aluminum and Copper (II) chloride
3CuCl2(aq) + 2Al(s) → 2AlCl3 + 3Cu(s)
A single replacement reaction is where an element replaces its like element in an ionic compound or acid. Metals always replace the cations and the nonmetals always replace the anions. The reaction will only occur if the element is more reactive than its like element in the compound. The aluminum and copper react to create a single replacement reaction. The aluminum, a metal, replaces the cation, copper (II), causing the aluminum foil to break down, as it forms aluminum chloride. Aluminum has a higher oxidation base, meaning it is more reactive, than the aqueous copper (II) chloride solution. This results in a reaction that breaks down the aluminum foil. When the foil is taken out of the circuit, the electricity takes the path through the light to light up the flare.
Chemical Change (Decomposition): hydrogen peroxide, potassium iodide, and dish soap
2H2O2(aq) → 2H2O(l) + O2(g)
The hydrogen peroxide is a decomposition reaction, where one reactant yields two or more products. The potassium iodide reacts with the hydrogen peroxide by breaking it down into oxygen and water. The dish soap captures the oxygen gas in bubbles, which also means a flame will grow when placed in the foam because of the oxygen gas stored in the bubbles. The foam is formed from the soap. Iodine is also produced, which is why the foam has a yellow tinge. This is an exothermic reaction where heat is released, so you may see steam and feel heat -- be careful!
Physical Change (Solid to Gas): dry ice, soap, and water
H2O + CO2 → H2CO3
The dry ice consists of frozen CO2. The water reacts with the ice, causing it to turn quickly into a gaseous form. This quick phase change causes a lot of steam to be released, creating the dense fog effect.
Alka-Seltzer Tablets, Water, and Oil
The alka-seltzer tablets are chemicals in solid form. When placed in the water, the alka-seltzer tablet dissolves due to the chemicals being released. It undergoes a reaction and changes from a solid to a liquid. Bubbles are produced. The oil swirls around and does not mix into the water because it is nonpolar. Since water is hydrophilic and oil is hydrophobic, they do not combine.
***DO NOT CONSUME ANY OF THE MATERIALS USED IN THESE EXPERIMENTS***
Wear protective gloves, goggles, and apron while all experiments are performed.
If chemicals contact your skin, rinse it in a nearby sink.
If chemicals get in your eyes, use a nearby fountain to flush it out.
If eyes or skin become irritated during experiment, rinse them with water.
If on fire or covered in large quantities of chemicals, use the safety shower.
Single Replacement Reaction: Wear protective gloves, apron, and goggles
- Small volumes of hydrogen gas are produced from the reaction - highly flammable
- keep flammable materials away from the reaction mixture.
- Copper (II) Chloride:
- Toxic when consumed
- Sodium bicarbonate:
- irritation in eyes
- Irritation of skin if water present
- Foam is very hot after reaction - do not touch, could get burned
- Hydrogen peroxide:
- Burns skin
- Strong oxidizing chemical - reacts dangerously with various substances
- Corrosive to eyes and skin
- potassium iodide:
- Irritates eyes and skin
- Dry ice:
- Extremely cold, could cause burns if contact too long - wear gloves
- Explosive if container does not allow for the release of gas
- Suffocation hazard due to large quantities of carbon gas - use ventilated area
This is the chemistry of all of our experiments within our board game:
Double Displacement Reaction: sodium bicarbonate (baking soda) and dilute acetic acid (vinegar)
NaHCO3(s) + CH3COOH(l) → CO2(g) + H2O(l) + Na+(aq) + CH3COO-(aq)
A double displacement reaction is where two aqueous ionic compounds exchange ions and form two new ionic compounds. It goes from two aqueous and aqueous to aqueous and either a solid, liquid, or gas. When this happens, it forms a precipitate. Acetic acid and sodium bicarbonate react in a double displacement reaction to form sodium acetate and carbonic acid. The carbonic acid then has a decomposition reaction and produces carbon dioxide gas which causes the solutions to bubble. Water and oxygen are also produced from this reaction. The release of the carbon dioxide gas flows out of the flask and into the balloon, inflating it.
Single Replacement Reaction: Aluminum and Copper (II) chloride
3CuCl2(aq) + 2Al(s) → 2AlCl3 + 3Cu(s)
A single replacement reaction is where an element replaces its like element in an ionic compound or acid. Metals always replace the cations and the nonmetals always replace the anions. The reaction will only occur if the element is more reactive than its like element in the compound. The aluminum and copper react to create a single replacement reaction. The aluminum, a metal, replaces the cation, copper (II), causing the aluminum foil to break down, as it forms aluminum chloride. Aluminum has a higher oxidation base, meaning it is more reactive, than the aqueous copper (II) chloride solution. This results in a reaction that breaks down the aluminum foil. When the foil is taken out of the circuit, the electricity takes the path through the light to light up the flare.
Chemical Change (Decomposition): hydrogen peroxide, potassium iodide, and dish soap
2H2O2(aq) → 2H2O(l) + O2(g)
The hydrogen peroxide is a decomposition reaction, where one reactant yields two or more products. The potassium iodide reacts with the hydrogen peroxide by breaking it down into oxygen and water. The dish soap captures the oxygen gas in bubbles, which also means a flame will grow when placed in the foam because of the oxygen gas stored in the bubbles. The foam is formed from the soap. Iodine is also produced, which is why the foam has a yellow tinge. This is an exothermic reaction where heat is released, so you may see steam and feel heat -- be careful!
Physical Change (Solid to Gas): dry ice, soap, and water
H2O + CO2 → H2CO3
The dry ice consists of frozen CO2. The water reacts with the ice, causing it to turn quickly into a gaseous form. This quick phase change causes a lot of steam to be released, creating the dense fog effect.
Alka-Seltzer Tablets, Water, and Oil
The alka-seltzer tablets are chemicals in solid form. When placed in the water, the alka-seltzer tablet dissolves due to the chemicals being released. It undergoes a reaction and changes from a solid to a liquid. Bubbles are produced. The oil swirls around and does not mix into the water because it is nonpolar. Since water is hydrophilic and oil is hydrophobic, they do not combine.
Here's how you play our game:
Guide:
Playing Instructions:
On each turn:
- Pink spaces - when you land on a pink space, draw a card from the Good Pile and follow the instructions on it, place the card at the bottom of the pile afterwards
- Red spaces - when you land on a red space, draw a card from the Bad Pile and follow the instructions on it, place the card at the bottom of the pile afterwards
- Orange Spaces - when you land on this space you will have to answer a riddle, if you answer it correctly move forward 4 places, if you answer it incorrectly move back 6 spaces
- Purple, green, and yellow spaces - these are free spaces, so you don’t need to do anything
Playing Instructions:
On each turn:
- Roll the dice
- Go the number of spaces indicated on the dice
- Follow the instructions according to the color of the space you land on (see instructions above)
Reflection: This project was actually really fun and I enjoyed it a lot. My group was really awesome and we all bounced ideas off of each other and collaborated on everything. One thing I think we did really well was sticking to one theme: desert. A couple of times we considered changing our theme but I'm glad we didn't because I think our theme was really unique and I doubt if we would have finished if we had changed themes. Another thing I think we did well was work supportively with one another. No one ever fought with each other and we all supported each other's ideas and helped them come to fruition. One thing I think we could've improved upon was our use of time. We spent a lot of days painting our board and making sure it looked good. However, this left not a lot of time to focus on the chemical reactions. We ended up getting them all done and being happy about it but it also left us with a lot of untested reactions that we didn't know how they would work when we actually tried them. I think personally I could have taken more intiative and helped more with the blueprint and safety precautions but other than that I think I did a good job of staying positive and supportive with my group.

